Metallurgical Engineering Gate Yearwise
Metallurgical Gate 2024
Metallurgical Gate 2023
Metallurgical Gate 2022
Metallurgical Gate 2021
Metallurgical Gate 2020
Metallurgical Gate 2019
Metallurgical Gate 2018
Metallurgical Gate 2017
Metallurgical Gate 2016
Metallurgical Gate 2015
Metallurgical Gate 2014
Metallurgical Gate 2013
Metallurgical Gate 2012
Metallurgical Gate 2011
Metallurgical Gate 2010
Metallurgical Gate 2009
Metallurgical Gate 2008
Metallurgical Gate 2007
Metallurgical Engineering Gate 2025 Questions with Answer
Ques 53 GATE 2025
Which of the following conditions is/are favorable for producing low-silicon hot metal in blast furnace ironmaking?
Ques 54 GATE 2025
Standard Gibbs free energies of formation of some solid oxides per mole of O2 at 1000 K are given below.
SiO2: -728 kJ, TiO2: -737 kJ, VO: -712 kJ, MnO: -624 kJ
Regarding thermodynamic feasibility of oxide reduction, which of the following statements is/are CORRECT under standard conditions at 1000 K?
Ques 55 GATE 2025
Consider the gas phase reaction: CO+(1/2)O2↔CO2. At equilibrium for a particular temperature, the partial pressures of CO, O2, and CO2 are found to be 10-6 atm, 10-6 atm, and 16 atm, respectively. The equilibrium constant for the reaction is ______ ×1010 (rounded off to one decimal place).
Ques 56 GATE 2025
Match the steel plant related processes in Column I with the associated information in Column II.
| Column I | Column II |
| P. Corex | 1. Melter-gasifier |
| Q. Electric Arc Furnace | 2. Natural gas reformer |
| R. Midrex | 3. Electromagnetic stirrer |
| S. Continuous Casting | 4. Hot heel |
Ques 57 GATE 2025
The excess molar Gibbs free energy of a solution of element A and B at 1000 K is given by GXS=-3000 XAXB J mol-1, where XA and XB are mole fractions of A and B, respectively.
The activity of B in a solution of A and B containing 40 mol% of B at 1000 K is ______ (rounded off to two decimal places).
Given: Ideal gas constant R=8.314 J mol-1K-1
Ques 58 GATE 2025
Molten steel at 1900 K having dissolved hydrogen needs to be vacuum degassed.
The equilibrium partial pressure of hydrogen to be maintained to achieve 1 ppm (mass basis) of dissolved hydrogen is ______ Torr (rounded off to two decimal places).
Given: For the hydrogen dissolution reaction in molten steel (1/2 H2(g)=[H]), the equilibrium constant (expressed in terms of ppm of dissolved H) is: log10Keq = -1900/T+2.4
1 atm = 760 Torr
Ques 59 GATE 2025
Consider the following reactions and their standard Gibbs free energies (in J):
Fe(s)+1/2 O2(g)↔FeO(s); ΔG°=-264900+65T
2 H2(g)+O2(g)↔2 H2O(g); ΔG°=-492900+109T
Assuming Fe and FeO to be pure and no solubility of gases in the solids, the value of pH2O/pH2 required to reduce solid FeO to solid Fe at 1000 K is ______ (rounded off to two decimal places).
Given: Ideal gas constant R=8.314 J mol-1K-1
Ques 60 GATE 2025
The standard Gibbs free energy change for the reaction Mg+Cd2+↔Mg2++Cd is ______ kJ (rounded off to an integer).
Given: Standard oxidation potentials for the reactions with respect to standard hydrogen electrode are:
Mg↔Mg2++2e-; E°=2.37 V
Cd↔Cd2++2e-; E°=0.403 V
Faraday's constant =96500 C mol-1
Ques 61 GATE 2025
Copper is being electrodeposited from a CuSO4 bath onto a stainless steel cathode of total surface area of 2 m2 in an electrolytic cell operated at a current density of 200 A m-2 with a current efficiency of 90%.
The mass of copper deposited in 24 h is ______ kg (rounded off to two decimal places).
Given: Faraday's constant =96500 C mol-1 atomic mass of copper =63.5g mol-1
Ques 62 GATE 2025
The reaction represented by A→B follows first order kinetics. At a given temperature, 20% of the reaction is completed in 223 s.
The time taken to complete 50% of the reaction at the same temperature is ______ s (rounded off to the nearest integer).
Ques 63 GATE 2025
The hydrostatic stress for the stress tensor provided below is ______ MPa (in integer).
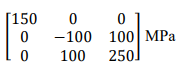
Ques 64 GATE 2025
For an isobaric process, the heat transferred is equal to the change in ______ of the system.
Ques 65 GATE 2025
Which one of the following is an intensive property?

Total Unique Visitors