Metallurgical Engineering > GATE 2025 > Phase Diagrams
Consider the phase diagram of a one component system given below. Vα, Vβ and VLiquid are the molar volumes of α, β, and liquid phases, respectively.
Which one of the following statements is TRUE?
Given: The change in molar enthalpies, ΔHα→β and ΔHβ→Liquid, are positive.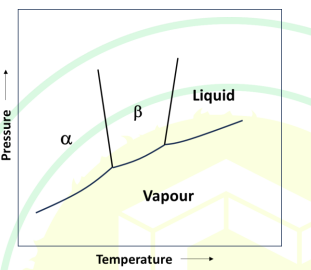
Which one of the following statements is TRUE?
Given: The change in molar enthalpies, ΔHα→β and ΔHβ→Liquid, are positive.

Correct : a
Similar Questions
800 grams of A-B alloy containing 20 wt.% B is held at temperature T1. The weight of B dissolved in α at that temperature is _______ grams. (Round off to...
In the Fe-C system, the invariant reaction Liquid + δ↔γ takes place at 1493°C. This type of reaction is called
Mr. X speaks _________ Japanese _________ Chinese.

Total Unique Visitors
Loading......