Metallurgical Engineering > GATE 2019 > Ellingham Diagram
The variation of standard free energies for two oxides AO and BO with temperature are shown below.
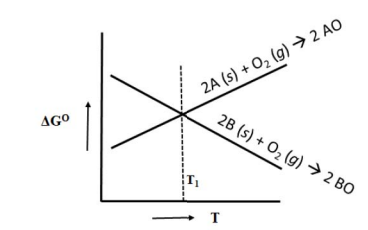 The correct statement with reference to the above figure is _______.
The correct statement with reference to the above figure is _______.

Correct : b
Similar Questions
Consider the following reactions and their standard Gibbs free energies (in J):Fe(s)+1/2 O2(g)↔FeO(s); ΔG°=-264900+65T2 H2(g)+O2(g)↔2 H2O(g)...
Mr. X speaks _________ Japanese _________ Chinese.
A sum of money is to be distributed among P, Q, R, and S in the
proportion 5 : 2 : 4 : 3, respectively.
If R gets ₹ 1000 more than S, what is the share of Q (...

Total Unique Visitors
Loading......