Civil Engineering > GATE 2020 SET-2 > Environmental Engineering
Alkalinity of water, in equivalent/litre (eq/litre), is given by 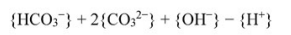 Where, {} represents concentration in mol/litre. For a water sample, the concentrations of HCO3- = 2 × 10-3 mol/litre, CO32- = 3.04 × 10-4 mol/litre and the pH of water = 9.0. The atomic weights are: Ca = 40; C = 12 and O = 16. If the concentration of OH- and H+ are NEGLECTED, the alkalinity of the water sample (in mg/litre as CaCO3), is
Where, {} represents concentration in mol/litre. For a water sample, the concentrations of HCO3- = 2 × 10-3 mol/litre, CO32- = 3.04 × 10-4 mol/litre and the pH of water = 9.0. The atomic weights are: Ca = 40; C = 12 and O = 16. If the concentration of OH- and H+ are NEGLECTED, the alkalinity of the water sample (in mg/litre as CaCO3), is
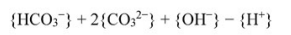
Correct : a
Similar Questions
The reaction rate involving reactants A and B is given by -k[A]α[B]β. Which one of the following statements is valid for the reaction to be a first-order reacti...
The wastewater from a city, containing a high concentration of biodegradable organics, is being steadily discharged into a flowing river at a location S. If the...
Which one of the following is NOT present in the acid rain?

Total Unique Visitors
Loading......