Chemical Engineering > GATE 2022 > Ideal Gas Processes
N moles of an ideal gas undergo a two-step process as shown in the figure. Let P, V, and T denote the pressure, volume, and temperature of the gas, respectively. The gas, initially at state-1 (P1, V1, T1), undergoes an isochoric (constant volume) process to reach state-A, and then undergoes an isobaric (constant pressure) expansion to reach state-2 (P2, V2, T2).
For an ideal gas, CP – CV = NR, where CP and CV are the heat capacities at constant pressure and constant volume, respectively, and assumed to be temperature independent. The heat gained by the gas in the two-step process is given by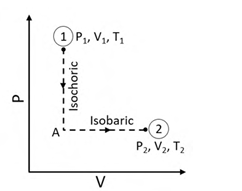

Correct : a
Similar Questions
Inhaling the smoke from a burning _________ could _________ you quickly.
A sphere of radius 𝑟 cm is packed in a box of cubical shape.
What should be the minimum volume (in cm3 ) of the box that can enclose the
sphere?
Pipes P and Q can fill a storage tank in full with water in 10 and 6 minutes,
respectively. Pipe R draws the water out from the storage tank at a rate of 34...

Total Unique Visitors
Loading......