Chemical Engineering > GATE 2022 > Enzyme Kinetics
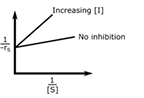
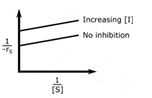
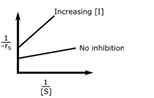
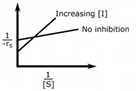
In an enzymatic reaction, an inhibitor (I) competes with the substrate (S) to bind with the enzyme (E), thereby reducing the rate of product (P) formation. The competitive inhibition follows the reaction mechanism shown below. Let [S] and [I] be the concentration of S and I, respectively, and rS be the rate of consumption of S. Assuming pseudo-steady state, the correct plot of
1⁄−rS vs 1⁄[S] is
Correct : a
Similar Questions
Consider an enzymatic reaction that follows Michaelis-Menten kinetics. Let KM. S, and Vmax denote the Michaelis constant, substrate concentration, and maximum r...
Inhaling the smoke from a burning _________ could _________ you quickly.
A sphere of radius 𝑟 cm is packed in a box of cubical shape.
What should be the minimum volume (in cm3 ) of the box that can enclose the
sphere?

Total Unique Visitors
Loading......