Chemical Engineering > GATE 2020 > Distillation
Equilibrium data for a binary mixture of E and F at two different pressures is shown in the figure.
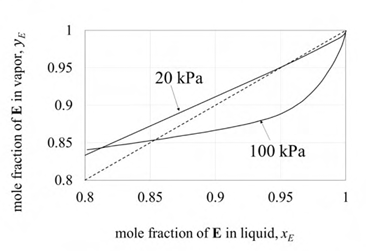 It is desired to process a feed containing 80 mol% E and 20 mol % F, and obtain a product with a purity of 99.5 mol % E. A sequence of two distillation columns, one operating at pressure P1 and another at P2, is employed for this operation, as shown below.
It is desired to process a feed containing 80 mol% E and 20 mol % F, and obtain a product with a purity of 99.5 mol % E. A sequence of two distillation columns, one operating at pressure P1 and another at P2, is employed for this operation, as shown below.
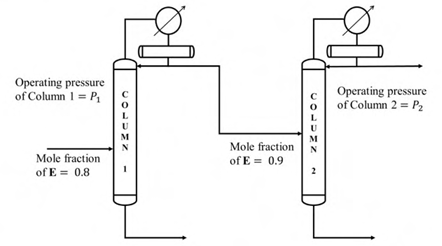 Mole fraction of E in the distillate obtained from column 1 is 0.9. If the column pressures P1 and P2 are in kPa, which one of the following is correct?
Mole fraction of E in the distillate obtained from column 1 is 0.9. If the column pressures P1 and P2 are in kPa, which one of the following is correct?


Correct : d
Similar Questions
A simple distillation column separates a binary mixture of A and B. The relative
volatility of A with respect to B is 2. The steady-state composition of A in t...
A packed distillation column, with vapor having an average molecular weight of 45 kg.kmol−1, density of 2 kg.m−3 and a molar flow rate of 0.1 kmol.s−1, has a fl...
Distillation of a non-reactive binary mixture with components 𝐴 and 𝐵 is carried
out in a batch still as shown in the figure below. The initial charge of the...

Total Unique Visitors
Loading......