Chemical Engineering > GATE 2020 > Absorption
SO2 from air is absorbed by pure water in a counter current packed column operating at constant pressure. The compositions and the flow rates of the streams are shown in the figure.
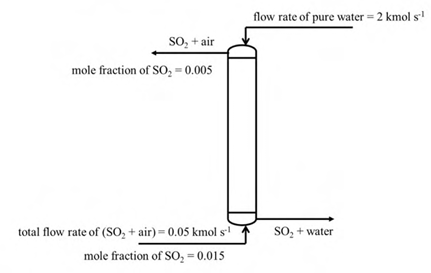 In addition, use the following data and assumptions
In addition, use the following data and assumptions
• Column operates under isothermal conditions
• At the operating temperature of the column, y*=40x, where y* is the mole fraction of SO2 in the gas that is in equilibrium with water containing SO2 at a mole fraction of x
• Solution is dilute and the operating line is linear
• Negligible amount of water evaporates
The number of transfer units (NTU) for this column is

• Column operates under isothermal conditions
• At the operating temperature of the column, y*=40x, where y* is the mole fraction of SO2 in the gas that is in equilibrium with water containing SO2 at a mole fraction of x
• Solution is dilute and the operating line is linear
• Negligible amount of water evaporates
The number of transfer units (NTU) for this column is
Correct : d
Similar Questions
A gas stream containing 95 mol% CO2 and 5 mol% ethanol is to be scrubbed with
pure water in a counter-current, isothermal absorption column to remove ethanol....
A liquid L containing a dissolved gas S is stripped in a countercurrent operation using a pure carrier gas V. The liquid phase inlet and outlet mole fractions o...
A countercurrent absorption tower is designed to remove 95% of component A from an incoming binary gas mixture using pure solvent B. The mole ratio of A in the...

Total Unique Visitors
Loading......