Chemical Engineering > GATE 2018 > Phase Equilibria
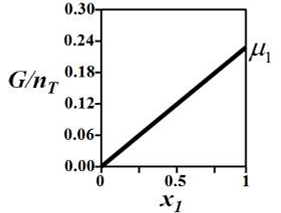
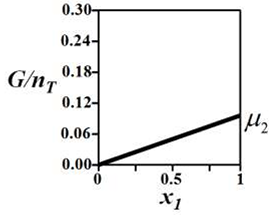
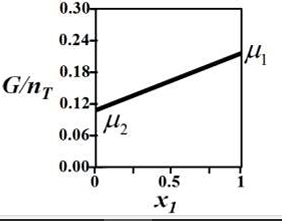
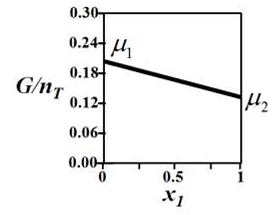
G denotes the Gibbs free energy of a binary mixture, nT denotes the total number of moles
present in the system, μi is the chemical potential of the ith component (μi ≠ μj and μi > 0), and xi is the mole fraction of the ith component.
The correct variation of G/nT (in J/mol) at constant temperature and pressure is given by __________.
present in the system, μi is the chemical potential of the ith component (μi ≠ μj and μi > 0), and xi is the mole fraction of the ith component.
The correct variation of G/nT (in J/mol) at constant temperature and pressure is given by __________.
Correct : c
Similar Questions
For a pure liquid, the rate of change of vapour pressure with temperature is 0.1 bar/K in the temperature range of 300 to 350 K. If the boiling point of the liq...
An azeotropic mixture of ethanol and water is to be separated in a distillation column using benzene as an entrainer. At the column operating conditions, two li...
Components A and B form an azeotrope. The saturation vapour pressures of A and B at the boiling temperature of the azeotrope are 87 kPa and 72.7 kPa, respective...

Total Unique Visitors
Loading......