Chemical Engineering > GATE 2017 > Compressibility Factor
The volumetric properties of two gases M and N are described by the generalized compressibility chart which expresses the compressibility factor (Z) as a function of reduced pressure and reduced temperature only. The operating pressure (P) and temperature (T) of two gases M and N along with their critical properties (Pc, Tc) are given in the table below.
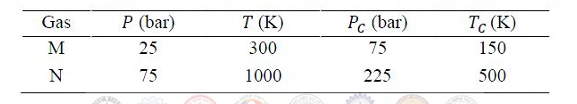 ZM and ZN are the compressibility factor of the gases M and N under the given operating conditions, respectively. The relation between ZM and ZN is
ZM and ZN are the compressibility factor of the gases M and N under the given operating conditions, respectively. The relation between ZM and ZN is

Correct : c
Similar Questions
Inhaling the smoke from a burning _________ could _________ you quickly.
A sphere of radius 𝑟 cm is packed in a box of cubical shape.
What should be the minimum volume (in cm3 ) of the box that can enclose the
sphere?
Pipes P and Q can fill a storage tank in full with water in 10 and 6 minutes,
respectively. Pipe R draws the water out from the storage tank at a rate of 34...

Total Unique Visitors
Loading......