Chemical Engineering > GATE 2012 > Material Balance with Recycle
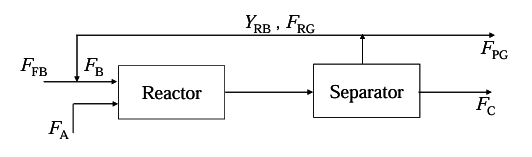
The reaction A(liq)+B(gas)→C(liq)+D(gas) is carried out in a reactor followed by a separator as shown below
 Notation:
Notation:
Molar flow rate of fresh B is FFB
Molar flow rate of A is FA
Molar flow rate of recycle gas is FRG
Mole fraction of B in recycle gas is YRB
Molar flow rate of purge gas is FPG
Molar flow rate of C is FC
Here, FFB=2 mol/s; FA=1 mol/s, FB/FA=5 and A is completely converted.
If the ratio of recycle gas to purge gas (FRG/FPG) is 4 then YRB is

Molar flow rate of fresh B is FFB
Molar flow rate of A is FA
Molar flow rate of recycle gas is FRG
Mole fraction of B in recycle gas is YRB
Molar flow rate of purge gas is FPG
Molar flow rate of C is FC
Here, FFB=2 mol/s; FA=1 mol/s, FB/FA=5 and A is completely converted.
If the ratio of recycle gas to purge gas (FRG/FPG) is 4 then YRB is
Correct : a
Similar Questions
Two elemental gases (A and B) are reacting to form a liquid (C) in a steady state process as per the reaction A+B→C. The single-pass conversion of the reac...
The reaction A(liq)+B(gas)→C(liq)+D(gas) is carried out in a reactor followed by a separator as shown below
Notation:Molar flow rate of fresh B is FFBMo...
Ammonia is synthesised at 200 bar and 773 K by the reaction N2+3H2↔2NH3. The yield of ammonia is 0.45 mol/mol of fresh feed. Flow sheet for the process (al...

Total Unique Visitors
Loading......