Chemical Engineering > GATE 2011 > Absorption
A gas mixture is in contact with a liquid. Component P in the gas mixture is highly soluble in the liquid. Possible concentration profiles during absorption of P are shown in the choices, where
x: mole fraction of P in bulk liquid
y: mole fraction of P in bulk gas
xi: mole fraction of P at the interface in liquid
yi: mole fraction of P at the interface in gas
y*: equilibrium gas phase mole fraction corresponding to xi
The CORRECT profile is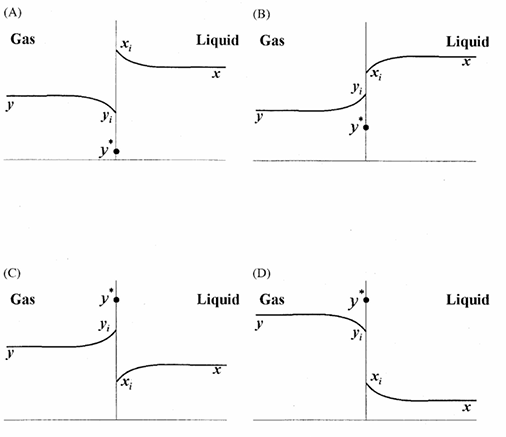
x: mole fraction of P in bulk liquid
y: mole fraction of P in bulk gas
xi: mole fraction of P at the interface in liquid
yi: mole fraction of P at the interface in gas
y*: equilibrium gas phase mole fraction corresponding to xi
The CORRECT profile is

Correct : a
Similar Questions
A gas stream containing 95 mol% CO2 and 5 mol% ethanol is to be scrubbed with
pure water in a counter-current, isothermal absorption column to remove ethanol....
A liquid L containing a dissolved gas S is stripped in a countercurrent operation using a pure carrier gas V. The liquid phase inlet and outlet mole fractions o...
A countercurrent absorption tower is designed to remove 95% of component A from an incoming binary gas mixture using pure solvent B. The mole ratio of A in the...

Total Unique Visitors
Loading......