Agricultural Engineering > GATE 2019 > Psychrometrics
Two streams of air with the following conditions are adiabatically mixed:
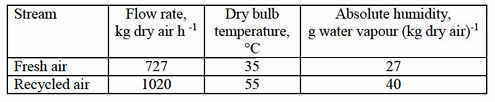 Latent heat of vapourization of water at 0 °C = 2501 kJ kg⁻¹
Latent heat of vapourization of water at 0 °C = 2501 kJ kg⁻¹
Specific heat capacity of dry air = 1.005 kJ kg⁻¹ K⁻¹
Specific heat capacity of water vapour = 1.880 kJ kg⁻¹ K⁻¹
Using above values, the dry bulb temperature and the absolute humidity of the mixed air in °C and g water vapour (kg dry air)⁻¹, respectively are

Specific heat capacity of dry air = 1.005 kJ kg⁻¹ K⁻¹
Specific heat capacity of water vapour = 1.880 kJ kg⁻¹ K⁻¹
Using above values, the dry bulb temperature and the absolute humidity of the mixed air in °C and g water vapour (kg dry air)⁻¹, respectively are
Correct : d
Similar Questions
Air-water vapour mixture at 30 °C DBT and 40% RH is heated to 65 °C DBT and 30 °C WBT and is used as drying medium under the constant rate period drying of spin...
Molecular masses of water and air are 18.02 and 28.97 kg (kg mol)⁻¹, respectively. Air in a room is at 40 °C under a total pressure of 101.3 kPa absolute and co...
Air-water vapour mixture at 1 atmosphere pressure has 0.035 kg water vapour (kg dry air)⁻¹ and dry bulb temperature of 37 °C. The value of Universal gas constan...

Total Unique Visitors
Loading......